CHEMISTRY
What Is The Definition Of Science?
I believe science is diving into the unknown and the experimentation of things to create new, official and exciting discoveries that have good, hard evidence to support your discovery.
Updated Version:
I believe science is the exploration and experimentation of thoughts and opinions to see what would be true and what would be false. There are many forms of science that can range from astronomy to food chemistry, but they are all related for one reason and that is to discover how everything works. I believe a vast majority of discoveries made in any field of science is important as long as those findings have been tested and are supported by reliable evidence. Sometimes knowledge comes at a price, for example the atom bomb was a very destructive weapon that could cause great damage if it or the knowledge to create it is in the wrong hands, so in a way you could say that knowledge is power, or with Adam and Eve who ate from the tree of knowledge of good and evil, they also gained knowledge but at a price.
What Is A Hypothesis And Why Would You Make One?
I believe hypothesis is a suggested explanation to answer a question based on reliable information I find it best as a simplified answer like nine instead of five, plus two, plus one, plus one making it easier to comprehend.
I believe science is diving into the unknown and the experimentation of things to create new, official and exciting discoveries that have good, hard evidence to support your discovery.
Updated Version:
I believe science is the exploration and experimentation of thoughts and opinions to see what would be true and what would be false. There are many forms of science that can range from astronomy to food chemistry, but they are all related for one reason and that is to discover how everything works. I believe a vast majority of discoveries made in any field of science is important as long as those findings have been tested and are supported by reliable evidence. Sometimes knowledge comes at a price, for example the atom bomb was a very destructive weapon that could cause great damage if it or the knowledge to create it is in the wrong hands, so in a way you could say that knowledge is power, or with Adam and Eve who ate from the tree of knowledge of good and evil, they also gained knowledge but at a price.
What Is A Hypothesis And Why Would You Make One?
I believe hypothesis is a suggested explanation to answer a question based on reliable information I find it best as a simplified answer like nine instead of five, plus two, plus one, plus one making it easier to comprehend.
Combustion
Combustion Article Annotations
Combustion is a chemical reaction that is rapid, self sustaining, and releases a reasonable amount of heat an example of combustion is a candle being lit and burning firewood. Three things are commonly present for combustion to occur:
Premixed Gas Flames
In this area of research, the fuel and oxidizer gases are mixed completely before ignition. Scientists are interested in the speed at which a flame zone travels away from the ignition source and into an unreacted mixture or flame speed as a function of both the oxidizer and fuel in an oxidizer-to-fuel ratio. If an oxidizer-to-fuel ratio is high enough or low enough the flame will not move into the unreacted mixture; these critical ratios are called upper and lower flammability limits which are of considerable interest regarding fundamental science and safety. Gravity strongly affect flammability limits and flame speed, mainly through buoyancy effects. Professionals researching in this field are also researching gravity's effects on the extinction, shape, structure, and stability of premixed gas flames.
Gaseous Diffusion Flames
In this area of research the oxidizer and fuel gases are originally separate. They tend to diffuse into each other and react upon ignition at their interface. The structure of these flames under the effects of microgravity is very different compared to if they were under the effects of earth's gravity because of buoyancy-induced flows caused by earth's gravity. Scientists study burning rates, flammability limits, and how diffusion flame structure affects soot formation. within this area, results from research on the behavior of gas-jet flames under the effects of microgravity, in both turbulent and transition flows, are being used to create models with potential applications in making strong strategies to control soot formation in many practical applications.
Liquid Fuel Droplets and Sprays
In this area of research, scientists study the combustion of individual fuel droplets suspended in an oxidizing gas. For these experiments, investigators mostly use fuels such as kerosene and methanol. Gravity impedes fundamental studies of droplet combustion under the effects of earth's gravity because of flows induced by high-density droplets that sink and buoyancy-induced upward acceleration of hot combustion products relative to the surrounding gas. These flows can cause drops to burn unevenly, making it harder for scientists to create meaningful conclusions from their experiments. This area of research also includes the investigation of the combustion of sprays and ordered arrays of fuel droplets under the effects of microgravity for a greater understanding of interactions between singular burning droplets in sprays. Knowledge from studies in this area of research should lead to improvements in the design of combustors using liquid fuels.
Fuel Particles and Dust Clouds
This area of research is particularly important when it comes to fire safety because clouds of coal dust are able to cause explosions in mines and grain-dust clouds are able to cause explosions in grain silos and grain elevators. It's especially difficult to research the fundamental characteristics of the combustion of fuel-dust clouds under the effects of normal gravity because originally well-dispersed dust clouds settle quickly because of the differences in density between the surrounding gas and the particles. Because during the sedimentation process particles tend to stick together and collide and because of that they form non-uniform fuel-ratios throughout the cloud. Fuel-dust clouds in microgravity remain evenly mixed , which allows scientists to gain more control over their experiments to reach a goal of reducing coal mine and grain elevator hazards.
Flame Spread Along Surfaces
Hindering flame spread along both solid and liquid surfaces is an important factor in fire safety. Flame spread involves the reaction between an oxidizer gas and the vapor that is made by "cooking" a condensed-phase fuel or a condensed phase fuel itself. Research has shown major differences in flame spread and ignition characteristics of liquid and solid fuels under the effects of microgravity and normal gravity. Results from material flammability tests done in 1 g, which are majorly impacted by buoyancy-induced flows, don't match the results obtained under the effects of microgravity. It's useful to study both material flammability and flame speed characteristics in microgravity to ensure safety in environments with different levels of gravity. What we learn from these studies will help us gain a better understanding of dangerous combustion reactions on earth. Experiments involving microgravity remove complexities involving buoyancy effects, providing a more fundamental scenario for creating flame-spreading theories.
Smoldering Combustion
Smoldering combustion is a fairly slow and non flaming combustion process involving a porous solid fuel and an oxidizer gas. An example of smoldering combustion is the "burning of a cigarette but it can happen on larger scales to, an example of that is with fuels like polyurethane foam. A porous fuel can create a large amount of gasified fuels if it has been smoldering for a long amount of time, which will be ready to react if an oxidizer flow occurs, which can encourage the fuel to create the transition to a full-fledged combustion mostly leading to catastrophic fires. The rate of combustion is fairly sensitive to heat exchange because heat is created slowly in this process; therefore, buoyancy effects are especially important. Accordingly, smoldering combustion is expected to act differently without any gravity.
Combustion Synthesis
This area of research is fairly new, it involves creating new materials through a combustion process and is connected to work in materials science. One area of reasonable interest is known as self-deflagrating high-temperature synthesis which happens when two materials (commonly solids) are mixed together, are reactive with each other, and create a reaction that creates an extreme amount of heat. When the reaction has begun, the flame will propagate through these particles in the form of a pressed mixture, resulting in creating a new material. A large amount of the opening research in this area includes changing variables such as preheat temperature, composition, and pressure. In the synthesis process, changing these factors leads to some interesting differences in the properties of materials. Fullerenes and nanoparticles can be created through flame processes. Fullerenes is a new form of carbon that is expensive to produce currently and cannot be produced in large amounts, but scientists predict that more uses for them will be produced as soon as there are readily available. Nanoparticles are also of amazing interest to materials scientists because of changes in the microstructure of compacted materials that can be made by sintering, which results in better properties of the final outcomes. These nanoparticles can then be used to create better pressed composite materials.
Hydrogen Balloon Hypothesis (Updated)
In chemistry we did an activity in which we mixed magnesium, citric acid, and water to make a chemical reaction where hydrogen was released into a balloon which we set aflame. What I think happened was that in the balloon the hydrogen atoms connected together to get eight electrons and the fire was enough to make the balloon pop and break up the hydrogen allowing the oxygen outside the balloon to connect with the hydrogen so then they can get 8 electrons allowing a brief chemical reaction creating H2O or water and a small explosion from the energy released when the atoms were connecting. I think this because what is required for a combustion reaction to happen you need a fuel, an oxidizer, and ignition, and without a single one of these factors the reaction won't happen, also we recently learned that in a reaction, atoms usually want to take and give electrons or combine so then they have a perfect eight electrons, releasing energy to create an explosion and that could lead to creating a different molecule such as H2O.
What is light?
For an experiment we did chemistry we were given a random chemical that we were supposed to burn over a candle, but the flame wasn't powerful enough to get the reaction that was wanted so the teacher got a blow torch and burned the chemicals himself, the chemical my group was given made the flame change color to a red. During and after this experiment we learned about the electromagnetic spectrum which is the way we categories forms of light from the largest wavelength to the shortest wave length, wave length is the distance between two peaks in a wave of light. We also learned about e-emission which is how flames can change color depending on what chemical is being burned, what happens during this process is that when a chemical or something else is being burned some of the electrons in their molecules gain energy and increase their orbit around the nucleus and then they release their energy in a form of light that could be a certain color depending on the wave length.
What Are The Phases Of Matter?
Pressure and temperature have the ability to change matter into different phases depending on how high or low both the temperature and pressure are. For example, recently in chemistry we observed that with enough pressure you are able to turn dry ice into a liquid and in releasing that pressure, you would create snow. The Phase Change Diagram is a diagram that shows at what points of pressure and temperature certain phases of matter will change into a solid, liquid, or gas depending on how high the temperature is and how much pressure is applied. During this activity we learned that it’s not only temperature that can change matter into different phases, it also depends on the pressure that's being inflicted upon matter. We are all aware that when you have a great enough heat then a solid will become a liquid, and with a greater heat a liquid will become a gas, during this activity we learned that it just so happens that with the right amount of pressure and heat can make a solid can skip that second phase of becoming a liquid and become a gas or a gas can skip that same phase and become a solid.
Properties of Matter
In chemistry we did another activity where we created balloons that we were able to keep at a neutral buoyancy out of trash bags, balsa wood, and a few candles. During this activity we learned about density, pressure, and buoyancy and how they effected the balloon that we made. Density is mass divided by volume and matter of lower density will float above matter of higher density and the way gas molecules move is by bouncing from object to object, by exposing that air to the heat the candles were creating were energizing the air molecules inside the trash bag, which caused the air molecules to move faster and force the trash bag to expand. Sometimes some air molecules come out of the bag, which would reduce the weight of the air inside the balloon and because the volume of the bag is also decreasing, the density of the bag is smaller than the density of the air around it, making the balloon have a positive buoyancy.
Phases of Matter 2: The Earth
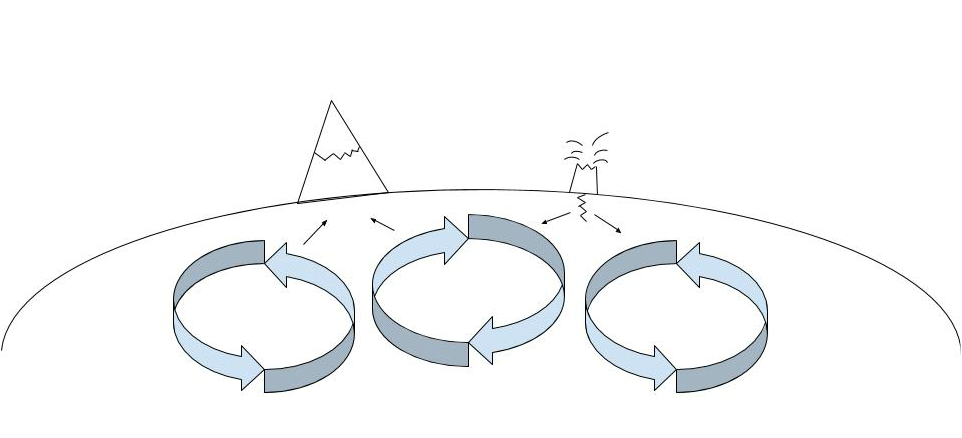
In chemistry we learned about how the earth functions and creates rocks by replicating that same process that happens in the earth in our class. We did this by mixing potassium chlorine and silicates, which is a similar process to how the earth mixes iron from the outer core (which is liquid and is mostly made up of iron and nickel) with silicates from the earth's mantle (which is liquid and is made up of about 97 percent silicates) through convection currents in the mantle. Convection currents go in a circular motion similar to the diagram I have posted below and have the ability to create mountains in areas where they are creating more pressure on the earth's surface, and volcanoes or fault lines when they are pulling away from each other. The rocks formed in such a way where they grew in vein-like branches that got thinner as the rocks grew, some of which were guided by small pockets of air.
Combustion is a chemical reaction that is rapid, self sustaining, and releases a reasonable amount of heat an example of combustion is a candle being lit and burning firewood. Three things are commonly present for combustion to occur:
- A fuel which could be solid like wood and coal, liquid like gasoline and kerosene, or a gas like propane and hydrogen.
- An oxidizer which could be a solid like potassium nitrate, liquid like hydrogen peroxide, or a gas like the air around us.
- A ignition stimulus which could be a fuse or an electric spark.
Premixed Gas Flames
In this area of research, the fuel and oxidizer gases are mixed completely before ignition. Scientists are interested in the speed at which a flame zone travels away from the ignition source and into an unreacted mixture or flame speed as a function of both the oxidizer and fuel in an oxidizer-to-fuel ratio. If an oxidizer-to-fuel ratio is high enough or low enough the flame will not move into the unreacted mixture; these critical ratios are called upper and lower flammability limits which are of considerable interest regarding fundamental science and safety. Gravity strongly affect flammability limits and flame speed, mainly through buoyancy effects. Professionals researching in this field are also researching gravity's effects on the extinction, shape, structure, and stability of premixed gas flames.
Gaseous Diffusion Flames
In this area of research the oxidizer and fuel gases are originally separate. They tend to diffuse into each other and react upon ignition at their interface. The structure of these flames under the effects of microgravity is very different compared to if they were under the effects of earth's gravity because of buoyancy-induced flows caused by earth's gravity. Scientists study burning rates, flammability limits, and how diffusion flame structure affects soot formation. within this area, results from research on the behavior of gas-jet flames under the effects of microgravity, in both turbulent and transition flows, are being used to create models with potential applications in making strong strategies to control soot formation in many practical applications.
Liquid Fuel Droplets and Sprays
In this area of research, scientists study the combustion of individual fuel droplets suspended in an oxidizing gas. For these experiments, investigators mostly use fuels such as kerosene and methanol. Gravity impedes fundamental studies of droplet combustion under the effects of earth's gravity because of flows induced by high-density droplets that sink and buoyancy-induced upward acceleration of hot combustion products relative to the surrounding gas. These flows can cause drops to burn unevenly, making it harder for scientists to create meaningful conclusions from their experiments. This area of research also includes the investigation of the combustion of sprays and ordered arrays of fuel droplets under the effects of microgravity for a greater understanding of interactions between singular burning droplets in sprays. Knowledge from studies in this area of research should lead to improvements in the design of combustors using liquid fuels.
Fuel Particles and Dust Clouds
This area of research is particularly important when it comes to fire safety because clouds of coal dust are able to cause explosions in mines and grain-dust clouds are able to cause explosions in grain silos and grain elevators. It's especially difficult to research the fundamental characteristics of the combustion of fuel-dust clouds under the effects of normal gravity because originally well-dispersed dust clouds settle quickly because of the differences in density between the surrounding gas and the particles. Because during the sedimentation process particles tend to stick together and collide and because of that they form non-uniform fuel-ratios throughout the cloud. Fuel-dust clouds in microgravity remain evenly mixed , which allows scientists to gain more control over their experiments to reach a goal of reducing coal mine and grain elevator hazards.
Flame Spread Along Surfaces
Hindering flame spread along both solid and liquid surfaces is an important factor in fire safety. Flame spread involves the reaction between an oxidizer gas and the vapor that is made by "cooking" a condensed-phase fuel or a condensed phase fuel itself. Research has shown major differences in flame spread and ignition characteristics of liquid and solid fuels under the effects of microgravity and normal gravity. Results from material flammability tests done in 1 g, which are majorly impacted by buoyancy-induced flows, don't match the results obtained under the effects of microgravity. It's useful to study both material flammability and flame speed characteristics in microgravity to ensure safety in environments with different levels of gravity. What we learn from these studies will help us gain a better understanding of dangerous combustion reactions on earth. Experiments involving microgravity remove complexities involving buoyancy effects, providing a more fundamental scenario for creating flame-spreading theories.
Smoldering Combustion
Smoldering combustion is a fairly slow and non flaming combustion process involving a porous solid fuel and an oxidizer gas. An example of smoldering combustion is the "burning of a cigarette but it can happen on larger scales to, an example of that is with fuels like polyurethane foam. A porous fuel can create a large amount of gasified fuels if it has been smoldering for a long amount of time, which will be ready to react if an oxidizer flow occurs, which can encourage the fuel to create the transition to a full-fledged combustion mostly leading to catastrophic fires. The rate of combustion is fairly sensitive to heat exchange because heat is created slowly in this process; therefore, buoyancy effects are especially important. Accordingly, smoldering combustion is expected to act differently without any gravity.
Combustion Synthesis
This area of research is fairly new, it involves creating new materials through a combustion process and is connected to work in materials science. One area of reasonable interest is known as self-deflagrating high-temperature synthesis which happens when two materials (commonly solids) are mixed together, are reactive with each other, and create a reaction that creates an extreme amount of heat. When the reaction has begun, the flame will propagate through these particles in the form of a pressed mixture, resulting in creating a new material. A large amount of the opening research in this area includes changing variables such as preheat temperature, composition, and pressure. In the synthesis process, changing these factors leads to some interesting differences in the properties of materials. Fullerenes and nanoparticles can be created through flame processes. Fullerenes is a new form of carbon that is expensive to produce currently and cannot be produced in large amounts, but scientists predict that more uses for them will be produced as soon as there are readily available. Nanoparticles are also of amazing interest to materials scientists because of changes in the microstructure of compacted materials that can be made by sintering, which results in better properties of the final outcomes. These nanoparticles can then be used to create better pressed composite materials.
Hydrogen Balloon Hypothesis (Updated)
In chemistry we did an activity in which we mixed magnesium, citric acid, and water to make a chemical reaction where hydrogen was released into a balloon which we set aflame. What I think happened was that in the balloon the hydrogen atoms connected together to get eight electrons and the fire was enough to make the balloon pop and break up the hydrogen allowing the oxygen outside the balloon to connect with the hydrogen so then they can get 8 electrons allowing a brief chemical reaction creating H2O or water and a small explosion from the energy released when the atoms were connecting. I think this because what is required for a combustion reaction to happen you need a fuel, an oxidizer, and ignition, and without a single one of these factors the reaction won't happen, also we recently learned that in a reaction, atoms usually want to take and give electrons or combine so then they have a perfect eight electrons, releasing energy to create an explosion and that could lead to creating a different molecule such as H2O.
What is light?
For an experiment we did chemistry we were given a random chemical that we were supposed to burn over a candle, but the flame wasn't powerful enough to get the reaction that was wanted so the teacher got a blow torch and burned the chemicals himself, the chemical my group was given made the flame change color to a red. During and after this experiment we learned about the electromagnetic spectrum which is the way we categories forms of light from the largest wavelength to the shortest wave length, wave length is the distance between two peaks in a wave of light. We also learned about e-emission which is how flames can change color depending on what chemical is being burned, what happens during this process is that when a chemical or something else is being burned some of the electrons in their molecules gain energy and increase their orbit around the nucleus and then they release their energy in a form of light that could be a certain color depending on the wave length.
What Are The Phases Of Matter?
Pressure and temperature have the ability to change matter into different phases depending on how high or low both the temperature and pressure are. For example, recently in chemistry we observed that with enough pressure you are able to turn dry ice into a liquid and in releasing that pressure, you would create snow. The Phase Change Diagram is a diagram that shows at what points of pressure and temperature certain phases of matter will change into a solid, liquid, or gas depending on how high the temperature is and how much pressure is applied. During this activity we learned that it’s not only temperature that can change matter into different phases, it also depends on the pressure that's being inflicted upon matter. We are all aware that when you have a great enough heat then a solid will become a liquid, and with a greater heat a liquid will become a gas, during this activity we learned that it just so happens that with the right amount of pressure and heat can make a solid can skip that second phase of becoming a liquid and become a gas or a gas can skip that same phase and become a solid.
Properties of Matter
In chemistry we did another activity where we created balloons that we were able to keep at a neutral buoyancy out of trash bags, balsa wood, and a few candles. During this activity we learned about density, pressure, and buoyancy and how they effected the balloon that we made. Density is mass divided by volume and matter of lower density will float above matter of higher density and the way gas molecules move is by bouncing from object to object, by exposing that air to the heat the candles were creating were energizing the air molecules inside the trash bag, which caused the air molecules to move faster and force the trash bag to expand. Sometimes some air molecules come out of the bag, which would reduce the weight of the air inside the balloon and because the volume of the bag is also decreasing, the density of the bag is smaller than the density of the air around it, making the balloon have a positive buoyancy.
Phases of Matter 2: The Earth
In chemistry we learned about how the earth functions and creates rocks by replicating that same process that happens in the earth in our class. We did this by mixing potassium chlorine and silicates, which is a similar process to how the earth mixes iron from the outer core (which is liquid and is mostly made up of iron and nickel) with silicates from the earth's mantle (which is liquid and is made up of about 97 percent silicates) through convection currents in the mantle. Convection currents go in a circular motion similar to the diagram I have posted below and have the ability to create mountains in areas where they are creating more pressure on the earth's surface, and volcanoes or fault lines when they are pulling away from each other. The rocks formed in such a way where they grew in vein-like branches that got thinner as the rocks grew, some of which were guided by small pockets of air.
Ocean Acidification
In class we talked about the chemistry behind (In my opinion) one of the world's major problems: ocean acidification. Ocean acidification happens when carbon dioxide dissolves into the ocean similar to how oxygen dissolves into the ocean which fish then extract from the water to breath. The carbon dioxide then mixes with H2O to create H2CO3 which is carbonic acid, after this process a hydrogen ion can break off to make a hydrogen ion and a bicarbonate ion or H+HCO3, after this another hydrogen ion can break off to make two hydrogen ions and a carbon trioxide ion or H2+CO3, and this is all in equilibrium meaning that either combination can transform into the other state that it is equilibrium to. CO3 is required to create the shells of animals such as pteropods and shellfish, when there is an increase in carbon dioxide in the ocean, there will be less CO3 because it will want to mix with the hydrogen that has been made in the process above to create carbonic acid.
Disappearing Ink Experiment
On November 17, 2016 we did an experiment where we created disappearing ink. To do this experiment yourself you would need to first measure 0.1 grams of phenophthalien into a 125 milliliter erlenmeyer flask. Next, add ten milliliters of ethanol and stir until the phenophthalien has dissolved. After that, add 100 milliliters of distilled water. Finally, add (drop wise) of NaOH with a molarity of 3 until the mixture has turned dark red. What is happening in this chemical reaction is that the CO2 from the air mixes with H2O to create H2CO3, after this a hydrogen ion can break off to make H+HCO3, after this another hydrogen ion can break off to make H2+CO3, which is similar to how ocean acidification occurs, and again this is all in equilibrium meaning that either combination can transform into the other state that it is equilibrium to. As the ink is is disappearing and the chemical reaction above is happening, the OH from the NaOH used in this mixture will mix with the hydrogen ions that were made in the process above to create H2O. In order to make the ink reappear, expose the ink to any base ammonia.
In class we talked about the chemistry behind (In my opinion) one of the world's major problems: ocean acidification. Ocean acidification happens when carbon dioxide dissolves into the ocean similar to how oxygen dissolves into the ocean which fish then extract from the water to breath. The carbon dioxide then mixes with H2O to create H2CO3 which is carbonic acid, after this process a hydrogen ion can break off to make a hydrogen ion and a bicarbonate ion or H+HCO3, after this another hydrogen ion can break off to make two hydrogen ions and a carbon trioxide ion or H2+CO3, and this is all in equilibrium meaning that either combination can transform into the other state that it is equilibrium to. CO3 is required to create the shells of animals such as pteropods and shellfish, when there is an increase in carbon dioxide in the ocean, there will be less CO3 because it will want to mix with the hydrogen that has been made in the process above to create carbonic acid.
Disappearing Ink Experiment
On November 17, 2016 we did an experiment where we created disappearing ink. To do this experiment yourself you would need to first measure 0.1 grams of phenophthalien into a 125 milliliter erlenmeyer flask. Next, add ten milliliters of ethanol and stir until the phenophthalien has dissolved. After that, add 100 milliliters of distilled water. Finally, add (drop wise) of NaOH with a molarity of 3 until the mixture has turned dark red. What is happening in this chemical reaction is that the CO2 from the air mixes with H2O to create H2CO3, after this a hydrogen ion can break off to make H+HCO3, after this another hydrogen ion can break off to make H2+CO3, which is similar to how ocean acidification occurs, and again this is all in equilibrium meaning that either combination can transform into the other state that it is equilibrium to. As the ink is is disappearing and the chemical reaction above is happening, the OH from the NaOH used in this mixture will mix with the hydrogen ions that were made in the process above to create H2O. In order to make the ink reappear, expose the ink to any base ammonia.